Intrauterine Devices (IUDs): Access and Coverage in the U.S.
Intrauterine devices (IUDs) are one of the most effective forms of reversible contraception. IUDs, along with implants, are known as long-acting reversible contraception (LARCs) because they can be used to prevent pregnancy for several years. IUDs have been used in the U.S. for decades, but a safety controversy in the 1970s prompted the removal of all but one IUD from the U.S. market by 1986. The first new generation IUD was introduced to the U.S. market in 1988, following revised Food and Drug Administration (FDA) safety and manufacturing requirements. Recent controversies have focused on the mechanism of action of IUDs, the high upfront costs for the device, and variability in insurance coverage and access. This fact sheet reviews FDA-approved IUDs, as well as use, availability, and key issues in insurance coverage and financing of IUDs in the U.S.
What is an IUD?
IUDs are small, T-shaped devices inserted into the uterus through the cervix by a trained medical provider to prevent pregnancy. A follow up visit may be recommended post-insertion to confirm its placement. IUDs are effective for three to 10 years and can be removed at any time up to the expiration date. There are two major categories of IUDs—copper and hormonal—and within those categories, there are currently six IUDs approved by the FDA (Table 1). In general, IUDs work by triggering an immune response in the uterus that prevents fertilization. Both copper and hormonal IUDs are more than 99% effective at preventing pregnancy. Like other forms of contraception, they do not protect against HIV and other sexually transmitted infections (STIs). IUDs do not affect an established pregnancy and do not act as an abortifacient.
Table 1
Types of Intrauterine Devices
| Non-hormonal | Available in the U.S. Since | Effectiveness | Use/Indications | Common Side Effects |
| Paragard Copper IUD | 1988 | 10 years | Can be used as emergency contraception when inserted within 5 days of unprotected sex | Prolonged and/or heavier menstrual bleeding, inter-menstrual spotting, higher frequency or intensity of cramps |
| Miudella Copper IUD* | Expected to be available in 2026 | 3 years | N/A | |
| Hormonal | Available in the U.S. Since | Effectiveness | Dosage and Indications | Common Side Effects |
| Mirena | 2001 | 5-8 years | 52mg; Can be used to treat heavy menstrual bleeding for up to 5 years and prevents pregnancy for up to 8 years | Inter-menstrual spotting, changes in menstrual bleeding Hormone-related headaches, nausea, breast tenderness, acne, mood changes, ovarian cysts, fatigue |
| Skyla | 2013 | 3 years | 13.5mg | |
| Liletta | 2015 | 5-8 years | 52mg; Can be used to treat heavy menstrual bleeding for up to 5 years and prevents pregnancy for up to 8 years | |
| Kyleena | 2016 | 5 years | 19.5mg |
Note: *The Miudella Copper IUD was approved for use in the U.S. in 2025 and is expected to be available to patients in 2026.
Non-Hormonal Copper-T Intrauterine Device
There are two copper IUDs available in the U.S.: Paragard and Miudella. Both are hormone-free, T-shaped devices wrapped in copper wire. Copper IUDs prevent pregnancy by affecting the function and mobility of sperm so that sperm cannot reach the uterus to fertilize an egg. Current evidence does not support prior theories that the copper IUD damages fertilized embryos or prevents implantation.
Paragard was first approved by the FDA in 1984 and has been available in the U.S. since 1988. Initially, this copper IUD was marketed by Teva Women’s Health Pharmaceuticals under the brand name ParaGard, but was acquired by CooperSurgical in 2017. Paragard is effective for up to 10 years and begins working immediately post-insertion, meaning it does not require the use of a backup method of contraception. Because of this, Paragard can also be used as emergency contraception within five days of unprotected intercourse or contraceptive method failure and is more effective at preventing pregnancy that emergency contraceptive pills. Unlike Plan B emergency contraceptive pills, the effectiveness of IUDs does not vary based on weight.
In 2025, the FDA approved the first new copper IUD in the U.S. in over 40 years, Miudella, from Sebela Pharmaceuticals. Miudella is effective for up to three years, but unlike Paragard, it has not been approved for use as emergency contraception. In addition to using less copper, the new device is smaller in size and is made of a flexible material called nitinol that allows for easier placement inside the uterus. Miudella is expected to be publicly available in the U.S. in the first half of 2026. Clinical trials evaluating its potential for long-term use are ongoing.
Hormonal Intrauterine Devices (LNG-IUD)
Four hormonal IUDs are currently available on the U.S. market: Mirena, Skyla, Liletta, and Kyleena. They are also known as LNG-IUDs because they contain the progestin hormone levonorgestrel, which is released into the body in small amounts each day to prevent pregnancy. Hormonal IUDs are not effective as emergency contraception and require the use of a backup method of contraception for the first seven days after insertion.
Mirena, manufactured by Bayer Healthcare Pharmaceuticals, is the most used hormonal IUD and has been available on the market longer than other hormonal IUDs. In addition to preventing pregnancy for up to eight years, the FDA approved the use of Mirena to treat heavy menstrual bleeding for up to five years.
Skyla, also manufactured by Bayer, is slightly smaller than the Mirena, making it a better candidate for people with a smaller uterus. It can prevent pregnancy for up to three years.
Liletta was developed by AbbVie and Medicines360—a non-profit women’s pharmaceutical company—specifically to be low cost and available to public health clinics enrolled in the national 340B Drug Pricing Program. The 340B program provides reduced cost pharmaceuticals to providersr that serve low-income populations.
Kyleena, the newest hormonal IUD, was approved by the FDA in September 2016 and became available in October 2016. It is also manufactured by Bayer and contains lower hormone levels than Mirena and Liletta.
Use, Awareness, and Availability of IUDs
Use of IUDs in the U.S. has been increasing substantially since the early 2000s but is still lower than other methods. Attitudes regarding the safety of IUDs have been shifting and interest has grown, especially among younger providers and contraceptive users who have less knowledge of the IUD controversies of the past.
Use
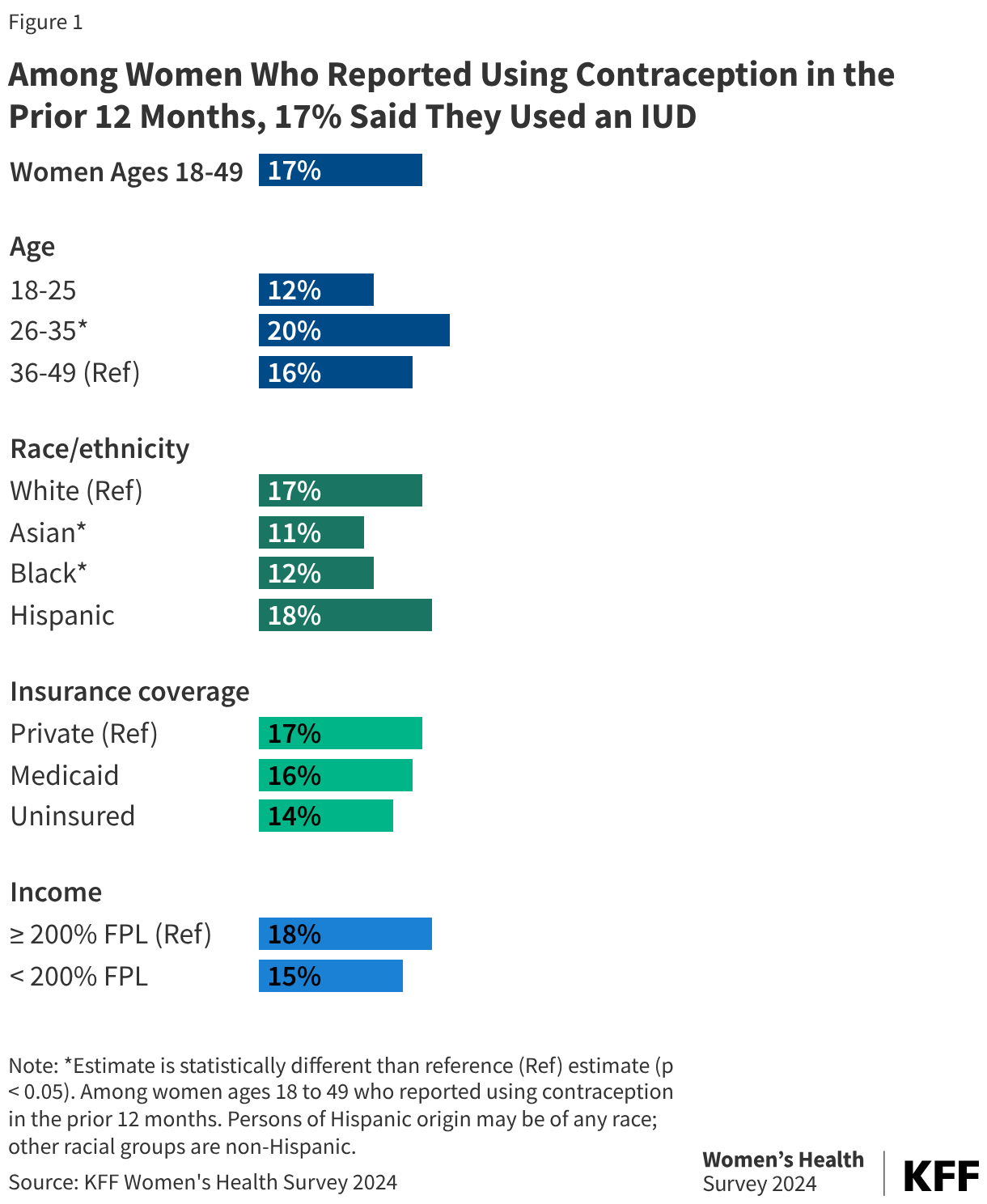
Data from the 2024 KFF Women’s Health Survey shows that 17% of women ages 18 to 49 years old who reported using contraception used an IUD in the last 12 months (Figure 1). IUD use is highest among women ages 26 to 35, compared to 12% among women ages 18-25, as well as Black women and Asian women (Figure 1). Multiple provider groups including the American College of Obstetricians and Gynecologists (ACOG) and the American Academy of Pediatrics (AAP) have recommended use of IUDs for all individuals, including adolescents, regardless of parity.
The promotion of LARCs as the “most effective” contraceptive methods has led to concerns about the potential for coercion, particularly due to racial biases in healthcare settings and the history of reproductive injustice against marginalized communities. Some people have reported that they have felt pressured to choose a method of contraception during contraceptive counseling and have felt that their providers preferred--and even pushed them towards--a LARC method because of their effectiveness in preventing pregnancy. Some individuals have reported that their physicians have been resistant and even unwilling to remove their IUDs early. Researchers have recommended that instead of first talking about LARCs and how effective they are, providers should first discuss with their patients their contraceptive preferences and reproductive goals and help patients choose a contraceptive method that meets their lifestyle needs.
Postpartum Use
Providing IUDs immediately following a delivery, miscarriage, or abortion is an effective strategy for averting unintended pregnancy in the postpartum period, with less than 1% of users getting pregnant within a year of insertion. This can also be convenient as it does not require scheduling separate appointments for IUD-related services. Some postpartum patients opt for a delayed insertion, meaning they get an IUD one to eight weeks after delivery, rather than immediate or early insertion. In a small study on patient preferences on postpartum LARC, patients cited concerns about expulsion rates and side effects as primary reasons for delaying IUD insertion. While studies have shown that expulsion rates of IUDs are higher with immediate postpartum insertion, results vary across studies and there may be several factors that influence the risk of expulsion during this time.
Social Media Influence
Some prominent social media influencers have utilized their social networks to share their negative experiences with and rationale for discontinuation of hormonal birth control methods such as oral contraceptive pills or, in the case of IUDs, they have sometimes focused on the pain of IUD insertion as well as side effects. Some patients have stated that clinicians do not pay enough attention to pain management, which may deter those interested in contraception from considering IUDs as an option. The most recent national contraception guidelines recommend administering lidocaine as a local anesthetic prior to IUD insertion to reduce patient pain, as there is significant evidence supporting the use of lidocaine for IUD insertions. In addition to these guidelines, it is also recommended that clinicians provide patients with thorough and individualized counseling related to anticipated pain.
Some social media influencers and organizations have made false claims about the harms and efficacy of hormonal contraception and have also conflated IUDs with abortion. Extensive research shows IUDs do not affect an established pregnancy.
Awareness and Availability
A 2023 KFF survey of OBGYNs found that nearly all OBGYNs provided IUDs in their practice (93%). However, some physicians require multiple visits for an IUD insertion, which can be inconvenient for patients with limited time and resources and may deter them from obtaining an IUD. While ACOG recommends OBGYNs facilitate same-day insertion procedures to improve patient access and experiences with contraceptive care, providers report encountering several barriers such as high upfront costs and challenges with storage and reimbursement processes for IUDs. Nonetheless, the share of publicly funded family planning clinics that perform same-day IUD insertions has increased overall. Between 2022 and 2023, about three in five (59%) clinics performed same-day insertions, compared to one in three (34%) in 2015.
Publicly funded family planning clinics are an important source of care for many low-income and uninsured people of reproductive age. Like other providers, access to IUDs has been challenging for some clinics due to high upfront costs, as well as limited training and staff capacity to provide IUDs. As a result, not all these sites provide the full range of contraceptive services to their patients including IUDs. Overall 71% of clinics offered hormonal IUDs while 64% offered copper IUDs from 2022 to 2023.
Insurance Coverage and Financing of IUDs
The costs of IUDs have been a barrier to their use for both patients and providers. Prices for an IUD can range from $0 to $1,800, in addition to provider visits for insertion, removal, and confirmation that the device was properly placed. The ACA’s contraceptive coverage requirement eliminated many out-of-pocket costs for many women, but some still do not have access to full coverage.
Private Insurance
The ACA includes a requirement that most private insurance plans must cover at least one type of all 18 FDA-approved contraceptive methods without cost sharing, meaning plans must cover the copper IUD and at least one hormonal IUD at no cost to policy holders. Plans determine which hormonal IUD they cover and must cover an alternate hormonal IUD if medically necessary. After the ACA contraceptive coverage mandate went into effect in 2012, studies found an increase in LARC initiation among women with private insurance coverage. Additional research found that the share of privately insured women paying $0 in out-of-pocket costs for IUDs doubled from 40% in 2012 to 88% in 2014. However, this analysis also suggests that the number of women with out-of-pocket expenses for IUD insertion increased between 2016 and 2020, despite the coverage requirement. While many women with private insurance (69%) continued to have no cost-sharing for IUD insertions in 2020, three in ten (31%) paid an average of $41 for IUD-related services.
Insurers can use medical management to help control costs and encourage beneficiaries to choose more affordable contraceptive methods. While insurers can require step therapy and prior authorization, federal guidance prohibits insurers from categorically restricting access to a method. Insurers can choose to cover generic contraceptives only while charging cost-sharing for the brand-name version, but since IUDs do not have a generic equivalent, the brand-name version must be covered without cost-sharing.
Medicaid
Federal law requires Medicaid programs to cover family planning services and supplies without cost-sharing, but there are variations in coverage between states and between different Medicaid populations. For enrollees of the traditional Medicaid programs that were in place prior to the passage of the ACA, coverage of IUDs is determined by each state program. States policies may limit coverage to only certain brands or types or apply medical management protocols to restrict availability.
Those who qualify for Medicaid under the ACA’s expansion of the program receive coverage for both the copper IUD and at least one hormonal IUD. The ACA requires these expansion programs to cover all FDA approved methods for women without cost-sharing, which is the same as the requirement for private insurance plans. Currently, 31 states and D.C. extend Medicaid coverage for family planning services, including contraception, to some uninsured women who do not qualify for full scope Medicaid.
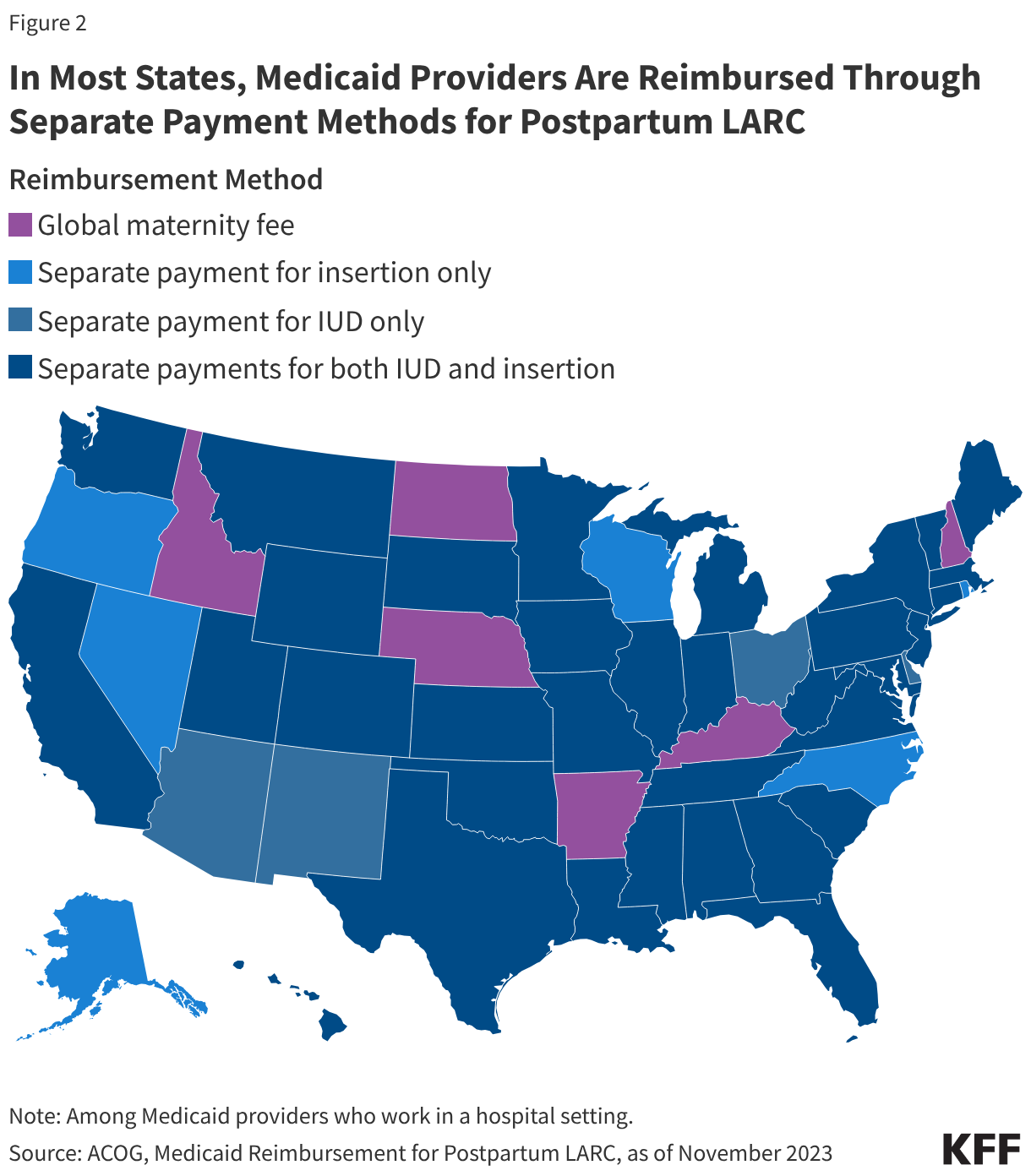
States have adopted a variety of payment policies to facilitate and improve access to postpartum LARC insertion. In the past, labor and delivery services were typically reimbursed through a single global fee. However, the global fee has previously been cited as a barrier to hospital LARC provision for not covering the costs of providing a LARC postpartum, and the absence of a separate fee or increase in reimbursement has become a disincentive for some providers. Over the last decade, most states have revised their Medicaid reimbursement policies to include a separate fee for postpartum LARC services, either a separate fee for LARC devices only, LARC insertion only, or separate fees for both (Figure 2). Six states—Idaho, North Dakota, Nebraska, Arkansas, Kentucky, and New Hampshire—continue to reimburse providers through a single global fee.
Uninsured
The federal Title X National Family Planning Program funds a network of clinics to provide family planning care to millions of low-income and uninsured people at reduced or no cost. Federal guidelines such as the CDC’s and the Office of Population Affairs’ Providing Quality Family Planning Services (QFP) and HRSA’s recommendations for preventive services for women state that offering women the full range of FDA-approved contraceptive methods is a critical element of quality family planning care.
Faced with higher costs or challenges getting reproductive care, uninsured women could decide to stop using contraception because they cannot afford it or switch to a less effective method, which could result in an increase of unwanted pregnancies and a loss of reproductive autonomy. The 2024 KFF Women’s Health Survey found one in five uninsured women of reproductive age has had to stop using a birth control method in the past 12 months because they couldn’t afford it, a rate that is four times greater than those with Medicaid (5%) or private insurance (2%). The uninsured rate is expected to increase substantially over the next decade due to forthcoming federal policy changes, particularly because of the 2025 federal budget law.
Community health centers (CHCs) and specialized family planning clinics such as Planned Parenthood centers play a major role in providing reproductive health care to low-income people, medically underserved communities, and people who are uninsured. CHCs are required to provide “voluntary family planning” services but have significant leeway in determining what specific services they provide. Research has found that sites that received Title X funds were consistently more likely to offer IUDs compared to sites that do not receive Title X funding. For example, nearly all (98%) of Planned Parenthood clinics offered same-day IUD insertions from 2022 to 2023, compared to just over a third (38%) of FQHCs.
Some manufacturers operate programs that offer reduced prices or fully subsidized IUDs for some low-income people. IUD manufacturers may also offer installment plans for those who purchase IUDs directly and have no other coverage.