A View from the States: Key Medicaid Policy Changes: Results from a 50-State Medicaid Budget Survey for State Fiscal Years 2019 and 2020
Benefits and Cost-Sharing
|
Key Section Findings
|
|
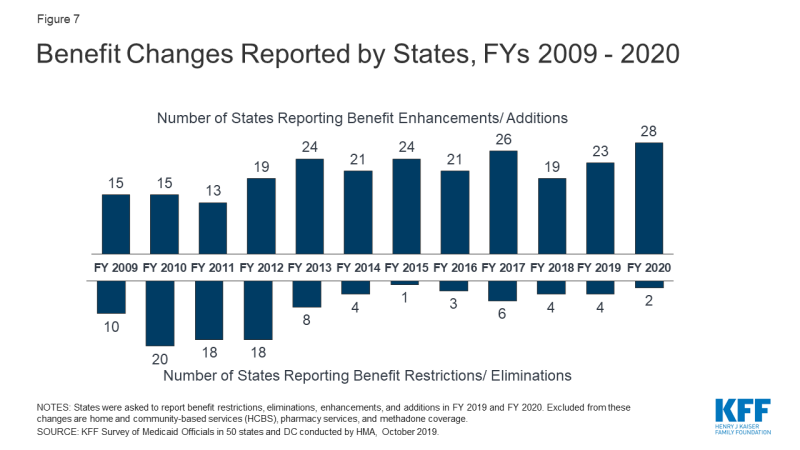
The number of states reporting benefit expansions (23 in FY 2019 and 28 in FY 2020) continues to significantly outpace the number of states reporting benefit restrictions (4 in FY 2019 and 2 in FY 2020). The most common benefit enhancements reported were for mental health/substance use disorder (SUD) services, but other service expansions include dental services, pregnancy and postpartum benefits, and diabetes prevention and care. Eleven states reported policies to eliminate or reduce a cost-sharing requirement for FY 2019 or FY 2020, exceeding the five states that reported new or increased cost-sharing requirements. States continue to pursue strategies to control high-cost prescription drugs and to address the opioid epidemic. What to watch:
Table 7 summarizes the nature of benefit policy changes by states in FY 2019 and FY 2020. |
Benefit Changes
The number of states reporting new benefits and benefit enhancements continues to significantly outpace the number of states reporting benefit cuts and restrictions. Twenty-three states reported new or enhanced benefits in FY 2019, and 28 states are adding or enhancing benefits in FY 2020. Few states reported benefit cuts or restrictions – four in FY 2019 and two in FY 2020 (Figure 7 and Table 7).
Similar to our findings in last year’s budget survey, many states reported expanding mental health and/or SUD services. Many of these mental health and SUD benefit expansions are incorporated into comprehensive Section 1115 waivers that include a request to use Medicaid funds for services provided in institutions for mental disease (IMDs) (more details about recent and planned IMD service changes and authorities used are discussed later in this section). Other non-IMD mental health and SUD service expansions that states reported include expanding access to crisis stabilization services and supporting recovery with new services such as peer supports. States also continue to increase access to medication-assisted treatment (MAT) services (detailed later in this section). Exhibit 14 highlights states implementing expanded mental health and/or SUD services and other select benefit enhancements.
States are also expanding pregnancy and postpartum services. For women covered by Medicaid under the pregnancy pathway, states are required to cover services “necessary for the health of a pregnant woman and fetus, or that have become necessary as a result of the woman having been pregnant.” Most states provide a comprehensive set of services for pregnant women.1 This year, states reported targeting additional pregnancy and postpartum services in FY 2019 and FY 2020. Some examples include the following: two states (Illinois2 and New Mexico) reported new home visiting programs; Georgia and New Jersey added CenteringPregnancy as a benefit to all or some members, a program designed to provide a set of prenatal services to Medicaid enrollees in a group setting; New Jersey and New York added doula service coverage; and Wyoming expanded coverage of midwife services. In addition, Missouri noted plans to seek approval through a demonstration waiver to add coverage of SUD treatment services and transportation for mothers who have given birth within the previous six months.
States are adding new benefits related to diabetes prevention and care. Diabetes Prevention Programs are evidence-based programs that aim to delay or prevent the onset of type 2 diabetes with targeted health behavior interventions. Two states (California, New Jersey) reported plans to cover Diabetes Prevention Program services in last year’s survey and these changes were implemented in FY 2019. Two more states (Maryland, New York) will begin covering Diabetes Prevention Program services effective in FY 2020. Additionally, South Carolina is expanding access to diabetes related care by covering continuous glucose monitoring devices for individuals with Type 1 diabetes and insulin-dependent gestational diabetes (FY 2020).
| Exhibit 14: Select Categories of Benefit Enhancements or Additions | ||||
| Benefit | FY 2019 | FY 2020 | ||
| Mental Health/SUD Services | 13 States | IL, MA, MD, NC, NH, NJ, NM, RI, SD, TX, UT, WA, WV | 20 States | AK, AL, CA, DC, DE, HI, IL, IN, KY, ME, MO, NE, NH, NV, OH, RI, TN, WI, WV, WY |
| Pregnancy and Postpartum Services | 4 States | GA, NJ, NM, NY | 4 States | IL, MO, NJ, WY |
| Dental Services | 4 States | IL, MA, MD, UT | 2 States | DE, VT |
| Diabetes Prevention and Care | 2 States | CA, NJ | 3 States | MD, NY, SC |
| Therapy Services (PT, OT, Speech) | 3 States | MO, NY, RI | 1 State | CA |
| Chiropractic Services | 2 States | DE, MO | 1 State | NE |
| Community Health Workers | 2 States | IN, SD | ||
| Telehealth Services | 1 State | TX3 | 2 States | MN,4 OH |
States reported initiatives to expand access to non-emergency medical transportation (NEMT). For example, Arizona added rideshare companies as NEMT providers and Colorado updated NEMT coverage policy to address urgent transportation needs by removing a requirement that rides be scheduled at least 48 hours in advance. In addition, the District of Columbia and Georgia are covering, or have plans to cover, “treatment without transport” services or transport to alternate locations by emergency medical services (EMS) transportation providers. These benefit changes are in line with the CMS Center for Medicare and Medicaid Innovation (CMMI) Medicare Emergency Triage, Treat, and Transport (ET3) payment model and August 2019 CMS guidance for reducing unnecessary transport to the emergency department in Medicaid programs.5
States also reported efforts to expand access to services for children. States are required to cover comprehensive services for children through the Early Periodic Screening, Diagnostic, and Treatment (EPSDT) benefit. Even with this comprehensive benefit, states reported an array of targeted service expansions for children including: intensive home-based services (Alabama, Illinois); trauma-informed care coordination for children with serious emotional disturbance (West Virginia); evidence-based parenting interventions (Maine); same day well-child and sick visits (South Carolina); and coverage of lead investigation services (North Carolina). States are also leveraging school settings to expand access to services for children. For example, in FY 2019 Texas expanded coverage of telemedicine services to occupational therapy and speech-language pathology provided in a school-based setting. In FY 2020, Ohio will expand coverage of telehealth services for behavioral health in schools, Michigan will expand provision of school-based services to general education students, and Nevada will extend coverage of school-based services to any community-based service provided in a school setting.
| SUPPORT Act Residential Pediatric Recovery Centers (RPRC) State Plan Options |
| In this year’s survey, states were asked whether they planned to adopt the SUPPORT Act’s state plan option to pay for services at residential pediatric recovery centers (RPRC) for services provided to infants under age 1 with neonatal abstinence syndrome (NAS) and their families. The rising incidence of NAS is associated with the nation’s opioid epidemic. Although infants with NAS have historically been treated in the hospital inpatient setting, states may be interested in other treatment settings like RPRCs. RPRCs can also provide services to mothers and other caretakers, including counseling or referrals for services, activities to encourage caregiver-infant bonding, and training on caring for infants with NAS.6
July 2019 CMS guidance7 indicates states may need to file a State Plan Amendment (SPA) to recognize RPRCs as a provider type and to update payment methodologies, as applicable. One state, Ohio, indicated it plans to adopt this option in FY 2020 joining West Virginia which, in FY 2018 and preceding the SUPPORT Act, became the first state to receive CMS SPA approval to finance NAS services using a bundled payment for providers outside of the hospital inpatient setting.8 The majority of states reported that they remain undecided about this opportunity or have no plans to file a SPA establishing RPRCs as a new provider type at this time. |
Most benefit restrictions in FY 2019 or FY 2020 are narrowly targeted. Benefit restrictions reflect the elimination of a covered benefit, benefit caps, or the application of utilization controls for existing benefits. This includes benefit restrictions that limit dental coverage (Alaska, Iowa) or implemented new prior authorization requirements (Colorado and Nevada9). In addition, Utah eliminated coverage of Early and Periodic Screening, Diagnostic, and Treatment (EPSDT) coverage for individuals ages 19 and 20 through a Section 1115 Waiver and Kentucky eliminated coverage of NEMT for methadone services for all non-pregnant adults and former foster youth (also through a Section 1115 waiver).
Cost-Sharing
Federal law limits cost-sharing for people with income below 100% FPL to “nominal” amounts (defined in federal regulations), with higher amounts allowed for beneficiaries at higher income levels. Total Medicaid premiums and cost-sharing for a family cannot exceed 5% of the family’s income on a quarterly or monthly basis.10 Certain groups are exempt from cost-sharing, including mandatory eligible children, pregnant women, most children and adults with disabilities, people residing in institutions, and people receiving hospice care. In addition, certain services are exempt from cost-sharing: emergency services, preventive services for children, pregnancy-related services, and family planning services.
Many state Medicaid programs require beneficiary cost-sharing, usually in the form of copayments, but to varying degrees. Research shows that even relatively small levels of cost-sharing in the range of $1 to $5 are associated with reduced use of care, including necessary services.11 Research also finds that cost-sharing can result in unintended consequences, such as increased use of the emergency room, and that cost-sharing negatively affects access to care and health outcomes.
In this survey, 15 states reported changes to cost-sharing requirements in either FY 2019 or FY 2020. Key changes are described below.
States were more likely to report policies to eliminate or reduce cost-sharing than report new or increased cost-sharing requirements. Eleven states reported policies to eliminate or reduce a cost-sharing requirement and five states reported new or increased cost-sharing in FY 2019 or FY 2020. Key changes include:
- Illinois, Montana, North Dakota, and New Mexico reported eliminating or plans to eliminate copayments on all services for some or all populations. For example, an approved Section 1115 waiver in New Mexico would have imposed a number of new copayments and other cost-sharing requirements, but the new administration did not implement the waiver’s changes and allowed existing copayments, including for people with disabilities, to sunset.
- The District of Columbia and Oklahoma are eliminating cost-sharing on medication-assisted treatment (MAT) and Michigan eliminated cost-sharing for all drugs used in the treatment of mental health conditions and SUD.
- Two states (Virginia and Wisconsin) reported new or increased copayments for non-emergency use of a hospital emergency department (ED) for certain populations.
- Kentucky reported changes that will prohibit MCOs from waiving copayments that apply in the Medicaid FFS program.
Pharmacy Cost Containment Actions in FY 2019 and FY 2020
As pharmacy expenditure growth became a greater Medicaid budget concern in the late 1990’s and early 2000’s, most states implemented an array of pharmacy cost containment strategies, including preferred drug lists (PDLs), supplemental rebate programs, state maximum allowable cost programs, multi-state purchasing pools, and prior authorization policies linked to clinical criteria. States continue to update and refine their drug utilization controls to respond to changes, especially new product offerings, in the pharmaceutical marketplace.
In this year’s survey, states were asked to describe any new or expanded pharmacy program cost containment strategies implemented in FY 2019 or planned for FY 2020. States were asked to exclude routine updates to PDLs or state maximum allowable cost programs as these utilization management strategies are employed by states regularly and are not typically considered major new policy initiatives. States reported a variety of actions in FY 2019 and FY 2020 to refine and enhance their pharmacy programs, often targeting new and emerging specialty and high-cost drug therapies, which many states noted as coming to the market with increasing speed and frequency.
Twenty-four states in FY 2019 and 26 states in FY 2020 reported newly implementing or expanding upon at least one initiative to contain costs in the area of prescription drugs. With growing political attention to the pharmaceutical supply chain, several states reported new and expanded initiatives to address concerns related to PBM transparency and the impact of spread pricing in managed care (at least seven states in both FY 2019 and FY 2020). A number of states also reported innovative purchasing arrangements, including value-based contracts linking pharmacy reimbursement to patient outcomes for certain high-cost drugs (three states in FY 2019 and five states in FY 2020). In addition, three states reported targeted initiatives specific to routine drugs used to treat chronic conditions like diabetes and asthma, including tightening monthly quantity limits on insulin and inhalers and instituting a new diabetes drug PDL.
Other strategies reported by states this year targeted uniform PDL requirements for MCOs. Four states report plans to implement a uniform PDL in FY 2020, and two additional states are expanding their uniform PDL policy in both FY 2019 and FY 2020. Additional highlights of state strategies to control pharmacy costs are noted below:
- Importation of prescription drugs. At the direction of the state legislature under Senate Bill 19-005, and subject to federal approval, Colorado plans to import prescription drugs from Canada.12 Under the Canadian Prescription Drug Importation Program, the Department of Health Care Policy and Financing will develop a wholesale drug importation list based on highest potential cost savings to the state and contract with one or more vendors to safely import and distribute drugs on the list.
- Direct negotiations with drug manufacturers. Beginning in FY 2020, Massachusetts will initiate direct negotiations with drug manufacturers with the goal of entering into supplemental rebate agreements for the highest cost drugs. If no agreement is reached, a public process will be used to determine the target value of the drug and improve transparency. Manufacturers may be referred to the state Health Policy Commission for further accountability. The state anticipates these reforms will save the Medicaid program $70 million in FY 2020.
- Subscription models. After obtaining final CMS approval in June 2019, Louisiana implemented a modified subscription model for hepatitis C antiviral therapies via a supplemental rebate agreement. The five-year partnership between the state and manufacturer set a capped expenditure amount, beyond which the state will continue to receive drugs at essentially no cost.
- Medicaid Drug Cap. New York continues to enhance its Medicaid Drug Cap as reported in last year’s survey. This initiative, which is separate from the state’s Medicaid Global Spending Cap, was initiated in FY 2018 and limits aggregate drug costs to an annual trend factor. For each year that costs exceed the allowable cap, the Department of Health negotiates enhanced rebates with the drug manufacturer and may refer drugs to the Drug Utilization Review (DUR) Board for additional review and recommendations as necessary.
- Prior authorization on certain new and/or high cost drugs. Four states used utilization management tools to address the unique challenges presented by new and/or high-cost drugs. For example, Kansas may place a temporary prior authorization requirement on new drugs that meet certain criteria until the state’s DUR Committee is able to adopt a more permanent policy. Nevada requires prior authorization for hemophilia drugs, oral oncology drugs, and drugs that exceed $10,000 per claim.
Strategies to Address Opioid Use Disorder
The opioid epidemic continues to impact individuals and communities across the country. According to the U.S. Department of Health and Human Services, as of 2016, 2.1 million people in the United States had an opioid use disorder and 11.5 million people misused prescription opioids as of 2016.13 The Centers for Disease Control and Prevention (CDC) indicates that drug overdose deaths are increasing nationally, the majority (around 68%) of which involve an opioid (prescription opioids, illicitly manufactured fentanyl and heroin).14 In 2017, 47,600 people died from an opioid overdose.15 The number of opioid overdose deaths is six times higher than it was in 1999, and approximately 130 people die from opioid-related drug overdoses each day.16
Medicaid plays a critical role in addressing the opioid epidemic, covering 4 in 10 people with opioid use disorder and expanding access to a range of treatment services.17 These services include inpatient detoxification, residential rehabilitation, outpatient detoxification, intensive outpatient services, and MAT medications. Many states have responded to July 2015 CMS guidance and November 2017 CMS guidance that allows states to seek Section 1115 waivers to pay for SUD services provided in IMDs.18,19 These state Medicaid director letters set out parameters for states to obtain Section 1115 waivers to try using federal Medicaid funds to provide short-term inpatient and residential SUD treatment services in IMDs. Signed into law October 24, 2018, the SUPPORT Act also includes a number of provisions related to Medicaid’s role in providing coverage and services to people who need opioid use disorder (OUD) treatment.20
Medicaid Pharmacy Benefit Management Strategies
This year’s survey asked states to report on specific Medicaid pharmacy benefit management strategies for preventing opioid-related harms that were in place in FY 2019 for FFS and changes to these strategies planned for FY 2020 (Exhibit 15). All states and the District of Columbia reported having at least one of these opioid-focused pharmacy management policies in FFS in place in FY 2019 and more than half of the states (32 states) plan to take further action in FY 2020. See Exhibit 15 and Table 8 for details on states implementing or expanding pharmacy benefit management strategies to reduce harm from opioid use or misuse.
| Exhibit 15: States Implementing Opioid-Focused Pharmacy Benefit Management Strategies in FFS | |||
| Strategy | In Place in FY 2019 (# of states) |
FY 2020 (# of states) | |
| New | Expanded | ||
| Adoption of opioid prescribing guidelines21 | 43 | 2 | 14 |
| Prospective drug utilization review22 | 46 | 0 | 17 |
| Prior authorization based on clinical criteria / step therapy23 | 49 / 36 | 0 / 0 | 16 / 6 |
| Retrospective drug utilization review24 | 48 | 0 | 15 |
| Medicaid prescribers must query state prescription drug monitoring program25 | 37 | 4 | 6 |
| Prescription drug lock-in programs26 | 44 | 0 | 6 |
States continue to report having point-of-sale safety edits, quantity limits, cumulative morphine milligram equivalents (MME) limits, and other utilization controls. Enhancements implemented in FY 2020 include adopting or updating opioid prescribing guidelines to align with guidance from the CDC, refining system edits (e.g., related to quantity, age, and/or concurrent use of opioids with benzodiazepines or antipsychotics), expanding prior authorization policy for both short- and long-acting opioids, and providing prescriber education and outreach based on retrospective drug utilization review activity. At least five states with MME limits reported lowering the threshold in FY 2020. Notable new initiatives include Florida’s new required urine drug screen for both initiation and continued opioid therapy and a new peer-to-peer prescriber outreach initiative in Utah focused on providers with high dose and high-risk prescribing patterns. Additionally, Delaware updated its prior authorization form with a recommendation to add naloxone to any opioid prescription with a dose in excess of 90 MME, and New York reported encouraging non-opioid pain management as part of its step therapy policy.
| SUPPORT Act Requirements & State Opportunities |
The SUPPORT Act requires state Medicaid programs to adopt by December 31, 2019 a variety of targeted pharmacy benefit management strategies to reduce opioid-related fraud, misuse, and abuse. As summarized in August 2019 CMS guidance,27 states must implement the following changes in their FFS delivery system and MCO contracts:
A number of states reported plans to make pharmacy benefit management changes to meet the requirements of the SUPPORT Act, but other states indicated they are already in compliance. The most commonly reported changes that will be made include implementation or expansion of point-of-sale safety edits, updated drug utilization review activities, and updating MCO contracts to include new requirements as applicable. The SUPPORT Act also directs states to implement Prescription Drug Monitoring Programs (PDMPs) and requires certain Medicaid providers to review the PDMP before prescribing a controlled substance by October 1, 2021. Thirty-seven states reported that they already have a legislative mandate or other policy that requires certain providers to check the state PDMP prior to prescribing opioids and four states are implementing this policy in FY 2020. One additional state (Montana) has a legislative mandate requiring prescribers to query the state PDMP beginning in FY 2021. Two states, Florida and Pennsylvania, report that review of the PDMP has been incorporated into the prior authorization process for prescribing opioids. The SUPPORT Act makes 100% FMAP available in FY 2019 and FY 2020 for certain PDMP implementation or connectivity activities, but only if the requesting state enters into agreements with border states to share PDMP data.28 In this year’s survey, we asked if states planned to access the 100% federal match for PDMP system design and development activities, such as connecting the PDMP to provider electronic health records (EHR) systems or performing other system upgrades. Most states reported that they remain undecided about this opportunity, but nine states indicate they used or plan to use the available enhanced federal match in FY 2019 and/or FY 2020. |
A majority of states that use MCOs to deliver pharmacy benefits require or partially require MCOs to follow the state’s FFS pharmacy benefit management policies for opioids. Fourteen states required MCOs to follow all of the state’s FFS pharmacy benefit management policies for opioids as of July 1, 2019, and 15 additional states reported MCOs must follow them in part. Of the 15 states with partial requirements, most reported some level of flexibility for MCOs to establish their own coverage criteria. At least four states require that MCOs be no more restrictive in their prior authorization or other criteria than FFS, while other states reported ongoing efforts to develop a more uniform opioid pharmacy management strategy across their FFS and managed care delivery systems.
Medication-Assisted Treatment coverage and access
The standard of care for opioid use disorder is MAT, which combines psychosocial treatment with medication.29 Compared to psychosocial treatment alone, MAT is associated with greater adherence to treatment, decreased opioid use, and reduced likelihood of overdose fatalities.30 There are three medications used as part of MAT for opioid use disorder: methadone, buprenorphine, and naltrexone (both oral and extended-release injectable forms).31,32 The SUPPORT Act requires state Medicaid programs to cover MAT, including all FDA-approved drugs,33 from October 2020 to September 2025. Today, all state Medicaid programs cover at least two MAT medications, and most cover all three.34
Most states cover methadone as FY 2019. In this year’s survey, forty-four states reported coverage of methadone in FY 2019, up from 38 states in FY 2018. Six states plan to add coverage for methadone in FY 2020 (Idaho, Kansas, Kentucky, Louisiana, Nebraska, and Tennessee). Wyoming reports plans to add methadone coverage prior to the October 1, 2020 deadline established by the SUPPORT Act, which falls in state fiscal year 2021. The state noted there are currently no certified methadone clinics operating in the state.
States experience a variety of challenges related to access to MAT. The most widely reported barrier to care is a shortage of waivered providers and rural providers in particular. States also identified shortages of behavioral therapy services, lack of knowledge of the evidence base for MAT, and provider stigma associated with substance use disorders as significant challenges. Other common challenges include lack of treatment resources for patients at all ASAM levels, low reimbursement rates, preference for the abstinence approach among providers, waivered-providers only accepting cash payment, and lack of access for pregnant women. In addition to these common challenges, other barriers identified include strict state licensing and registration requirements, difficulties for primary care providers to integrate MAT into daily practice routines, lack of transportation, limited MAT access in jails and prisons, failure to incorporate MAT into clinical training programs, few telehealth options, and waivered providers not treating beneficiaries with OUD.
Many states reported removing or relaxing prior authorization requirements to expand access to MAT. In this year’s survey, we asked states to describe initiatives or policies implemented in FY 2019 or planned for FY 2020 to address MAT access challenges. Twenty-one states indicated changes in prior authorization requirements to improve access to MAT in FY 2019 and FY 2020. Three states reported adding vivitrol to the PDL and/or expanding MAT coverage policy to include vivitrol. Vivitrol is an extended-release injectable form of naltrexone that can be prescribed by any healthcare provider licensed to prescribe medications.35
In addition to reducing administrative barriers like prior authorization, a number of states report multi-faceted initiatives to expand access to MAT. Some of these initiatives are funded through State Targeted Response to the Opioid Crisis (STR) and State Opioid Response (SOR) grants. Activities include collaborating with emergency departments, prisons, and other community partners; leveraging telehealth capabilities; opening new treatment centers; increasing MAT reimbursement rates; and supporting providers with training and other resources. Examples of state strategies to expand access to MAT include:
- Using STR and SOR grants, Arizona opened six 24/7 Opioid Treatment on Demand Centers, four in metropolitan areas and two in rural areas. The Medicaid program has also increased the number of peer support specialists in the state and launched a “feet-on-the-street” outreach campaign to identify and engage high-risk beneficiaries and refer them to OUD treatment resources. Additional efforts include helping finance an OUD /MAT stigma reduction campaign through the Governor’s Office and providing MAT information sessions for professional associations, universities, and substance abuse coalitions.
- The Office of Behavioral Health (OBH) in Colorado implemented the IT MATTTRs2 initiative which offers incentives to providers to obtain a Drug Enforcement Authority (DEA) waiver to prescribe MAT. This initiative also delivers team training and practice supports to make sure providers have tools and resources to adopt a comprehensive MAT program.
- Massachusetts and New York require administration of buprenorphine to individuals presenting in the emergency department with an OUD. The District of Columbia implemented a MAT induction pilot that screens emergency department patients for SUD risk and connects at-risk patients to a peer recovery coach to discuss initiation of MAT. Interested patients will begin buprenorphine in the emergency department and receive a warm handoff to community services within 48 hours. Demonstration projects in South Carolina involve participation by five anchor hospitals where emergency department staff identify patients in need of MAT and refer for coordination and ongoing treatment through community partners.
- Arizona and Illinois operate a peer consultation line for providers with questions regarding MAT therapies.
- New Jersey initiated a modified hub and spoke model Office Based Addiction Treatment program that offers clinical support and enhanced reimbursement for physicians that provide care management services in addition to MAT. It is also requiring residential programs to offer MAT, as an alternative to an abstinence-only approach and is in the process of increasing after-hours access to MAT to support working individuals and late releases from jail.
Institutions for Mental Disease (IMD) Flexibilities
In an effort to address the opioid epidemic as well as broader behavioral health initiatives, CMS and Congress have acted to give states additional flexibility to provide services in residential treatment facilities and other settings that would otherwise qualify as “institutions for mental disease,” or IMDs, and be ineligible for federal Medicaid funding. States have been actively using these new flexibilities to expand the availability of substance use disorder and mental health services for Medicaid beneficiaries. According to this year’s survey, a large majority of states (43 states)36 report they plan to use at least one of the following flexibilities to provide services in IMDs in FY 2020:
- Managed Care “In Lieu of” Authority. The 2016 Medicaid Managed Care Final Rule allows states, under the authority for health plans to cover services “in lieu of” those available under the Medicaid state plan, to receive federal matching funds for capitation payments on behalf of nonelderly adults who receive inpatient psychiatric or SUD treatment or crisis residential services in an IMD for no more than 15 days during a given month.37
- Section 1115 Waivers. CMS guidance issued under the Obama Administration in July 2015 and later revised by the Trump Administration in November 2017 allows states to obtain Section 1115 waivers of the federal IMD payment exclusion as part of a broader demonstration to improve access to a continuum of treatment services for opioid use disorder (OUD) and other substance use disorders.38,39,40 The guidance sets out goals and milestones states would need to meet as part of their Section 1115 demonstration in order to receive federal Medicaid funds for short-term inpatient and residential SUD treatment services in IMDs. In November 2018, CMS issued new guidance inviting states to apply for Section 1115 waivers to receive funding for services provided in an IMD for adults with a serious mental illness (SMI) or children with a serious emotional disturbance (SED) as part of an effort to expand services across the care continuum.41 States must meet milestones regarding care quality, care coordination, increased access to crisis stabilization services, and earlier identification and engagement in treatment. This guidance reverses prior CMS policy to not use Section 1115 waiver authority to allow Medicaid payments for non-elderly adults with a primary mental health diagnosis in IMDs.
- The SUPPORT Act. In October 2018, President Trump signed into law, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act.42 The legislation created a new state plan option, from October 1, 2019 to September 30, 2023, to cover IMD services for up to 30 days in a year for individuals with an SUD. The SUPPORT Act also codified the 2016 Medicaid Managed Care final rule provision allowing MCOs to offer “in lieu of” IMD coverage for up to 15 days in a month and authorizes Medicaid payments for services provided outside IMDs for pregnant and postpartum women receiving IMD SUD services, as of October 2018.43
On this year’s survey, states were asked about whether they currently use or plan to use the managed care “in lieu of” authority (MCO states only), plan to pursue the Section 1115 SMI/SED waiver opportunity, or plan to adopt the SUPPORT Act state plan option:
More than three-quarters of MCO states (31 states) reported using “in lieu of” authority in their managed care programs in both FY 2019 and FY 2020 (Table 9). Two states reported plans to begin using this authority in FY 2020 and two states reported using this authority in FY 2019 but not in FY 2020.44 Just four MCO states indicated they did not use “in lieu of” authority in their managed care contracts in either year.
One state submitted in FY 2019 and 12 states plan to submit (in FY 2020 or after) Section 1115 IMD waivers for services for individuals with SMI or SED (Table 9).45 Nine states indicated they do not plan to pursue a SMI/SED IMD waiver while 28 states reported their plans were undetermined at this time.46 In addition, as of October 2019, 26 states had an approved IMD SUD waiver while 3 states had pending waiver requests at CMS (not shown in table). (States were not asked on this survey about plans to submit IMD SUD waivers; however, IMD SUD pending/approved waiver activity is tracked separately).47
Only five states reported plans to pursue the SUPPORT Act IMD SPA option in FY 2020 (Table 9). Twenty-one states reported they have not yet determined if they will pursue the SUPPORT Act IMD SPA option and 24 states indicated they do not plan to adopt this option.48 In explaining the rationale for not pursuing this opportunity, many states noted they had a Section 1115 waiver in place or were in the process of implementing a Section 1115 waiver that would allow for funding of IMD services, removing the need to pursue the SPA option. Several states also felt that the Section 1115 waiver would provide more flexible limits on length of IMD stays compared to the 30-day cap on IMD services under the SPA option.
Table 7: Benefit Changes in all 50 States and DC, FY 2019 and FY 2020
|
States |
FY 2019 |
FY 2020 |
||
|
Enhancements/ Additions |
Restrictions/ Eliminations |
Enhancements/ Additions |
Restrictions/ Eliminations |
|
|
Alabama |
X |
|||
|
Alaska |
X |
X |
||
|
Arizona |
X |
|||
|
Arkansas |
||||
|
California |
X |
X |
||
|
Colorado |
X |
X |
||
|
Connecticut |
||||
|
Delaware |
X |
X |
||
|
DC |
X |
X |
||
|
Florida |
||||
|
Georgia |
X |
|||
|
Hawaii |
X |
|||
|
Idaho |
||||
|
Illinois |
X |
X |
||
|
Indiana |
X |
X |
||
|
Iowa |
X |
|||
|
Kansas |
||||
|
Kentucky |
X |
X |
||
|
Louisiana |
||||
|
Maine |
X |
|||
|
Maryland |
X |
X |
||
|
Massachusetts |
X |
|||
|
Michigan |
X |
|||
|
Minnesota |
X |
|||
|
Mississippi |
X |
X |
||
|
Missouri |
X |
X |
||
|
Montana |
||||
|
Nebraska |
X |
|||
|
Nevada |
X |
X |
||
|
New Hampshire |
X |
X |
||
|
New Jersey |
X |
X |
||
|
New Mexico |
X |
|||
|
New York |
X |
X |
||
|
North Carolina |
X |
|||
|
North Dakota |
||||
|
Ohio |
X |
|||
|
Oklahoma |
||||
|
Oregon |
||||
|
Pennsylvania |
||||
|
Rhode Island |
X |
X |
||
|
South Carolina |
X |
|||
|
South Dakota |
X |
|||
|
Tennessee |
X |
|||
|
Texas |
X |
|||
|
Utah |
X |
X |
||
|
Vermont |
X |
|||
|
Virginia |
||||
|
Washington |
X |
|||
|
West Virginia |
X |
X |
||
|
Wisconsin |
X |
|||
|
Wyoming |
X |
|||
|
Totals |
23 |
4 |
28 |
2 |
|
NOTES: States were asked to report benefit restrictions, eliminations, enhancements, and additions in FY 2019 and FY 2020. Home and community-based services (HCBS) and pharmacy benefit changes are excluded from this table. Methadone benefit changes were also excluded from this table. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2019. |
||||
Table 8: Medicaid FFS Pharmacy Benefit Management Strategies for Opioids in all 50 States and DC, in Place in FY 2019 and Actions Taken in FY 2020
|
States |
Opioid Prescribing Guidelines |
Prospective DUR |
Prior Authorization and Step Therapy |
Retrospective DUR |
Required use of Prescription Drug Monitoring Programs |
Prescription Drug Lock-In Programs |
Any Opioid Management Strategies |
|||||||
|
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
In place FY 2019 |
New/Exp FY 2020 |
|
|
Alabama |
X |
X+ |
X |
X |
X |
|||||||||
|
Alaska |
X |
X |
X |
X+ |
X+ |
X |
X |
X |
X |
X |
X |
X |
||
|
Arizona |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
Arkansas |
X |
X |
X |
X |
X |
X |
X |
|||||||
|
California |
X |
X |
X |
X |
X |
|||||||||
|
Colorado |
X |
X |
X |
X+ |
X+ |
X |
X |
X |
X |
X |
||||
|
Connecticut |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|||
|
Delaware |
X |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
||||
|
DC |
X |
X |
X |
X |
X |
X |
||||||||
|
Florida |
X |
X |
X |
X+ |
X+ |
X |
X |
X |
X |
X |
||||
|
Georgia |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
Hawaii |
X |
X |
||||||||||||
|
Idaho |
X |
X |
X+ |
X |
X* |
X |
X |
X |
||||||
|
Illinois |
X |
X |
X |
X |
X |
X |
X |
|||||||
|
Indiana |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
X |
X |
||
|
Iowa |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
|||||
|
Kansas |
X |
X+ |
X |
X |
||||||||||
|
Kentucky |
X |
X |
X+ |
X |
X |
X |
||||||||
|
Louisiana |
X |
X |
X |
X |
X |
X |
X |
|||||||
|
Maine |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
X |
|||
|
Maryland |
X |
X |
X |
X |
X |
X |
||||||||
|
Massachusetts |
X |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
X |
X |
|
|
Michigan |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
X |
X |
||
|
Minnesota |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
||||
|
Mississippi |
X* |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
||||
|
Missouri |
X |
X |
X |
X+ |
X |
X |
X |
X |
||||||
|
Montana |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
|||||
|
Nebraska |
X |
X+ |
X |
X |
X |
X |
X |
X |
||||||
|
Nevada |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|||||
|
New Hampshire |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
New Jersey |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|||
|
New Mexico |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
New York |
X |
X |
X |
X |
X+ |
X+ |
X |
X |
X |
X |
X |
X |
||
|
North Carolina |
X |
X |
X |
X+ |
X+ |
X |
X |
X |
X |
X |
X |
X |
||
|
North Dakota |
X |
X+ |
X |
X |
X |
X |
||||||||
|
Ohio |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
|||||
|
Oklahoma |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
X |
||||
|
Oregon |
X* |
X |
X |
X |
X |
X |
X |
X |
X |
X |
X |
|||
|
Pennsylvania |
X |
X |
X |
X |
X |
X |
X |
|||||||
|
Rhode Island |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
|||||
|
South Carolina |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
South Dakota |
X |
X |
X |
X+ |
X |
X |
X |
|||||||
|
Tennessee |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
Texas |
X |
X |
X |
X |
X |
X* |
X |
X |
||||||
|
Utah |
X |
X |
X |
X+ |
+ |
X |
X |
X |
X |
X |
X |
|||
|
Vermont |
X |
X |
X |
X+ |
X |
X |
X |
X |
X |
X |
||||
|
Virginia |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
Washington |
X |
X |
X |
X |
X |
X |
X* |
X |
X |
X |
||||
|
West Virginia |
X |
X |
X+ |
X |
X |
X |
X |
|||||||
|
Wisconsin |
X |
X |
X+ |
X |
X |
X |
X |
X |
||||||
|
Wyoming |
X |
X+ |
X |
X* |
X |
X |
X |
|||||||
|
Totals |
43 |
16 |
46 |
17 |
49 |
17 |
48 |
15 |
37 |
10 |
44 |
6 |
51 |
32 |
|
NOTES: States were asked to report whether they had select pharmacy benefit management strategies in place in their FFS programs in FY 2019, and/or had plans to adopt or expand these strategies in FY 2020. Prospective drug utilization review activies includes screening prescription drug claims, while retrospective drug utilization review examines already-paid prescription drug claims. “*” indicates that a policy was newly adopted in FY 2020, meaning that the state did not have any policy in that category/column in place in FY 2019. “+” indicates step therapy policies. Utah responded that it plans to expand step therapy in FY 2020, but not prior authorization. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2019. |
||||||||||||||
Table 9: New IMD Service Authorities and Actions Taken or Planned by States
|
States |
States using the Medicaid managed care “in lieu of” authority for enrollees |
States pursuing a Section 1115 IMD waiver for services for individuals with SMI or SED+ |
States planning to adopt the SUPPORT Act State Plan option# |
|||||||
|
Yes – in FY 2019^ |
Yes – in FY 2020 |
Yes – in both FY 2019 and FY 2020 |
Undetermined |
Submitted in FY 2019 |
Plan to submit in FY 2020 |
Plan to Submit after FY 2020 |
Undetermined |
Plan to Adopt in 2020 |
Undetermined |
|
|
Alabama |
— |
— |
— |
— |
X |
X |
||||
|
Alaska |
— |
— |
— |
— |
X |
X |
||||
|
Arizona |
X |
X |
||||||||
|
Arkansas |
X |
X |
X |
|||||||
|
California |
X |
|||||||||
|
Colorado |
X |
|||||||||
|
Connecticut |
— |
— |
— |
— |
X |
|||||
|
Delaware |
X |
X |
||||||||
|
DC |
X |
X |
||||||||
|
Florida |
X |
X |
X |
|||||||
|
Georgia |
X |
X |
X |
|||||||
|
Hawaii |
X |
X |
X |
|||||||
|
Idaho |
— |
— |
— |
— |
X |
X |
||||
|
Illinois |
X |
X |
||||||||
|
Indiana |
X |
X |
X |
|||||||
|
Iowa |
X |
|||||||||
|
Kansas |
X |
X |
X |
|||||||
|
Kentucky |
X |
X |
X |
|||||||
|
Louisiana |
X |
|||||||||
|
Maine |
— |
— |
— |
— |
X |
|||||
|
Maryland |
NR |
NR |
NR |
NR |
NR |
NR |
||||
|
Massachusetts |
X |
X |
X |
|||||||
|
Michigan |
X |
X |
||||||||
|
Minnesota |
X |
X |
X |
|||||||
|
Mississippi |
X |
|||||||||
|
Missouri |
X |
X |
||||||||
|
Montana |
— |
— |
— |
— |
X |
X |
||||
|
Nebraska |
X |
X |
||||||||
|
Nevada |
X |
X |
X |
|||||||
|
New Hampshire |
X |
X |
X |
|||||||
|
New Jersey |
X |
X |
||||||||
|
New Mexico |
X |
X |
X |
|||||||
|
New York |
X |
X |
X |
|||||||
|
North Carolina |
— |
X |
X |
|||||||
|
North Dakota |
||||||||||
|
Ohio |
X |
X |
||||||||
|
Oklahoma |
— |
— |
— |
— |
X |
X |
||||
|
Oregon |
X |
|||||||||
|
Pennsylvania |
||||||||||
|
Rhode Island |
X |
X |
||||||||
|
South Carolina |
X |
X |
||||||||
|
South Dakota |
— |
— |
— |
— |
X |
|||||
|
Tennessee |
X |
X |
X |
|||||||
|
Texas |
X |
X |
||||||||
|
Utah |
X |
X |
||||||||
|
Vermont |
— |
— |
— |
— |
X |
X |
||||
|
Virginia |
X |
X |
X |
|||||||
|
Washington |
X |
X |
||||||||
|
West Virginia |
X |
X |
X |
|||||||
|
Wisconsin |
X |
X |
X |
|||||||
|
Wyoming |
— |
— |
— |
— |
X |
X |
||||
|
Totals |
2 |
2 |
31 |
2 |
1 |
8 |
4 |
28 |
5 |
21 |
|
NOTES: NR – not reported. “–” indicates state without MCOs. States without an “x” on a given row under each column, indicated “no plans to adopt/submit.” *The 2016 Medicaid Managed Care Final Rule allows states, under the authority for health plans to cover services “in lieu of” those available under the Medicaid state plan, to receive federal matching funds for capitation payments on behalf of adults who receive inpatient psychiatric or SUD treatment or crisis residential services in an IMD for no more than 15 days during a given month. +In November 2018, CMS issued new guidance inviting states to apply for Section 1115 waivers of the federal IMD payment exclusion for services for adults with serious mental illness (SMI) or children with serious emotional disturbance (SED). #The SUPPORT Act legislation created a new state plan option, from October 1, 2019 to September 30, 2023, to cover IMD services for up to 30 days in a year for individuals with an SUD. Re: adoption of SUPPORT Act SPA option, states were also given the response option, “plan to submit after FY 2020.” No states selected this option. ^These states (NM, WV) may have discontinued use of Medicaid managed care “in lieu of” authority in FY 2020 due to approval/implementation of Section 1115 IMD SUD waivers. SOURCE: Kaiser Family Foundation Survey of Medicaid Officials in 50 states and DC conducted by Health Management Associates, October 2019. |
||||||||||